|
The quantity denoted by D, the Debye unit, is still commonly used to express dipole moments. The nunber of valence electrons in oxygen is 6.Īnother way,carbon is an element of group 4A,so it has four valence electrons.On the other hand, oxygen is an element of group 6A,so it has six valence electrons.\ C \cdot m = 4.8 \ D\] To get the valence electrons of oxygen,we need to look at the electronic configuration of oxygen. The nunber of valence electrons in carbon is 4.Īgain, we have to find out the valence electrons of oxygen. In the free carbon monoxide molecule, a net negative charge remains at the carbon end and the molecule has a small dipole moment of 0.122 D. The top left corner of this structure, labeled 5, prime, shows a phosphorus atom single bonded to three oxygen atoms, one of which has a superscripted negative charge, and double bonded to a fourth oxygen atom. In contrast to intramolecular forces, such as the covalent bonds that hold atoms together in molecules and polyatomic ions, intermolecular forces hold molecules together in a liquid or solid. Prolonged exposure to carbon monoxide rich atmospheres may be fatal. The properties of liquids are intermediate between those of gases and solids, but are more similar to solids. 
Agency for Toxic Substances and Disease Registry (ATSDR) Carbon monoxide is a colorless, odorless gas. It is found in both outdoor and indoor air. The highest value of principal quantum number ,n, indicates the valence shell and we know the electrons in valence shell is called valence shell. Carbon monoxide is a colorless, nonirritating, odorless, and tasteless gas. The highest value of principal quantum number here is n=2. The carbon and oxygen atom have unequal charge distribution and therefore CO bond has a net dipole moment making CO a polar molecule. Find more Chemistry widgets in WolframAlpha. 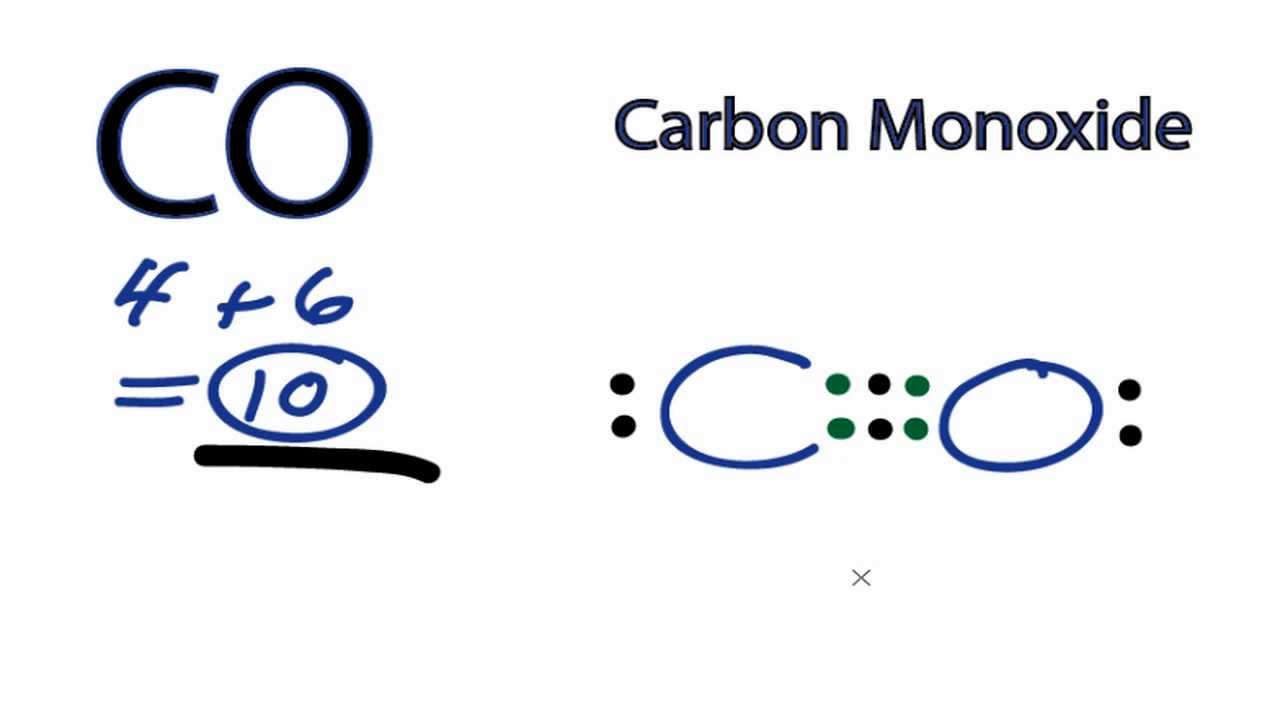
Get the free 'Lewis Structure Finder' widget for your website, blog, Wordpress, Blogger, or iGoogle. To get the valence electrons of carbon,we need to look at the electronic configuration of carbon. CO (Carbon monoxide) is polar in nature because of the difference in electronegativity of carbon (2.55) and oxygen (3.44) atoms. The molecular shape of H 2 CO is -linear bent or angular/ trigonal planar/ tetrahedral/ trigonal pyramid. This widget gets the Lewis structure of chemical compounds. There is no net molecule dipole moment since the. In comparison, CO2 does not have a dipole moment because it is linear and the bond dipoles cancel each other out. 
The oxygen will 'pull' on the carbon creating a dipole moment. D) all of the above are true E) none of the above, The Lewis structure for carbon monoxide is : C O : This structures shows A) 2 lone pairs and 1 bonding pair. CO can be linear and have a dipole moment because it is not cancelling out. 
To draw the CO Lewis structure, we have to find out the CO valence electrons first.We express valence electrons as dots in lewis dot structure. Since carbon C and oxygen O have different electronegativities, electrons arent shared equally between them. Study with Quizlet and memorize flashcards containing terms like When a nonmetal bonds with a nonmetal A) a molecular compound forms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
